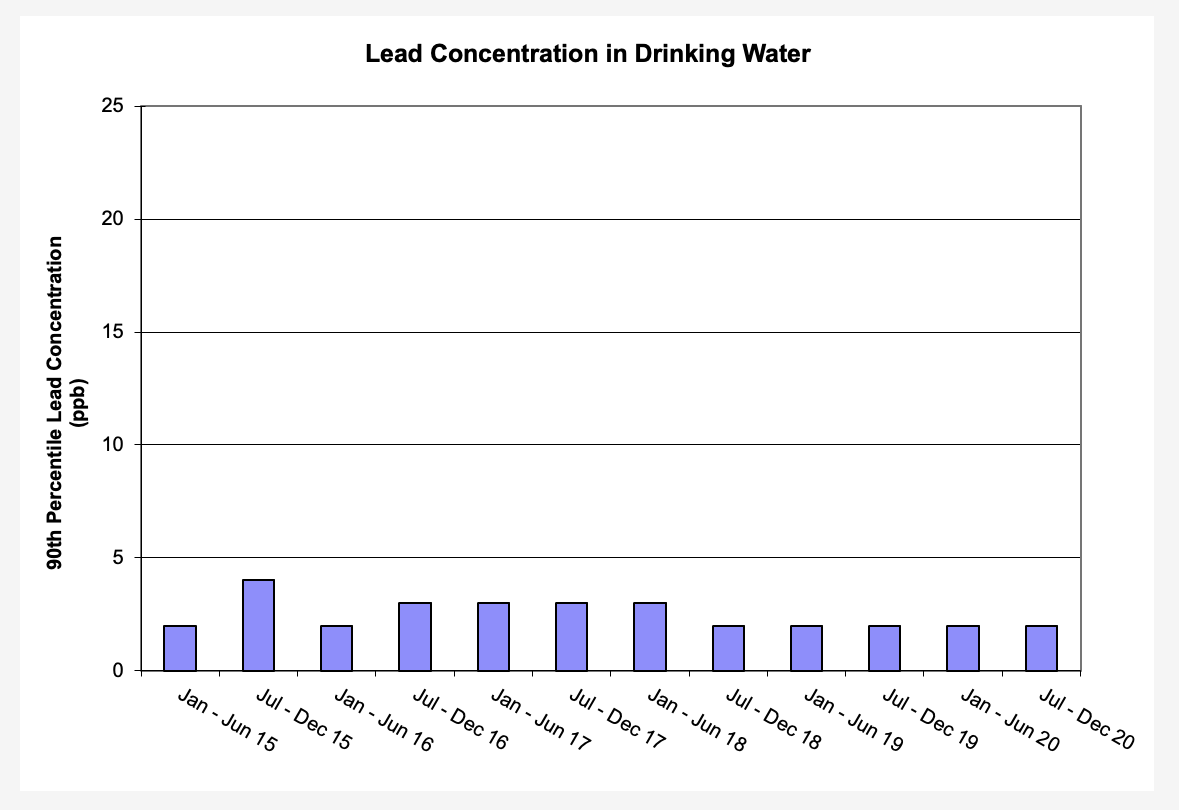
Corrosion control treatment is used to prevent pipe corrosion and the presence of metals in drinking water. Drinking water treatment plants such as the Washington Aqueduct add orthophosphate, a corrosion control treatment, before water leaves the treatment plant. Orthophosphate creates a thin protective coating inside pipes and plumbing fixtures and is very effective in reducing the presence of lead and other metals in drinking water.
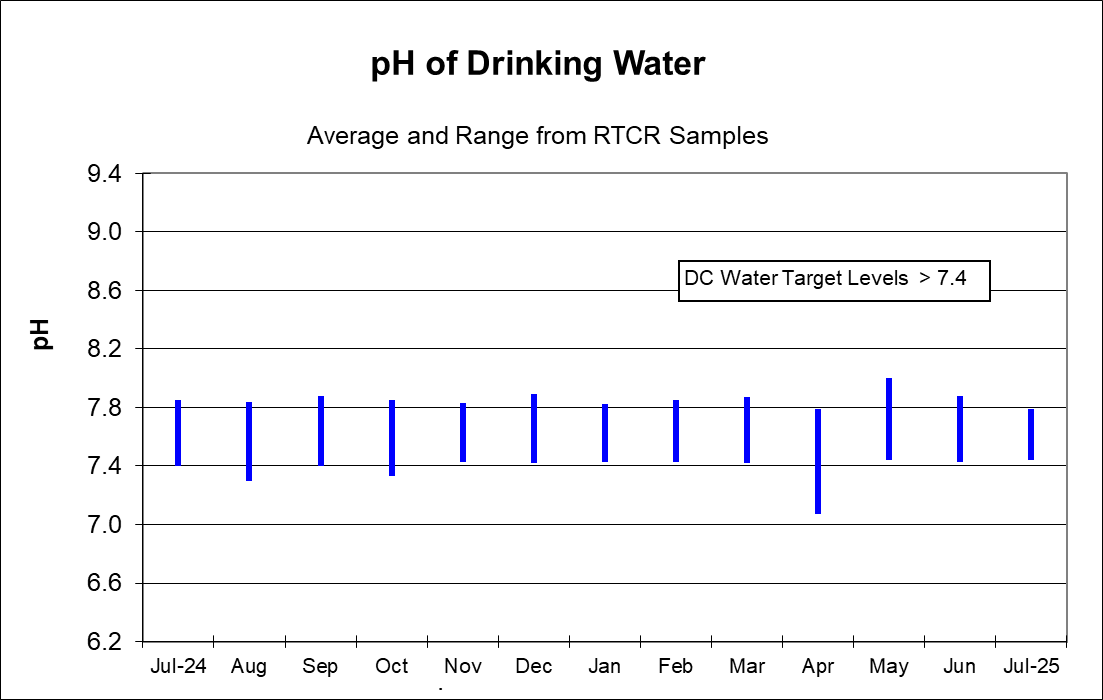
Water can become corrosive at lower pH values. The pH level of drinking water reflects how acidic it is. Water pH is measured on a scale of 0 to 14, with 7 considered neutral, meaning there’s a balance between the water’s acidity and basicity. Raising the pH of drinking water makes the water less corrosive which also strengthens an existing protective coating on the interior of the pipe. That coating reduces the likelihood of lead getting into the water as it passes through customer-owned water service lines, household plumbing and faucets that contain lead.
The corrosion treatment chemical added to the water, orthophosphate, works best at a pH range of 7.0 to 8.0. The Washington Aqueduct is required to maintain a pH between 7.4 and 8.0 for water leaving the treatment plant. DC Water monitors the pH in the distribution system daily to ensure these levels stay within the optimum range for orthophosphate. DC Water has set a target range of 1.0 to 4.5 mg/L orthophosphate to target in the distribution system.
These Tests Confirm our Corrosion Control Treatments Work
The graphs below shows the maximum, average, and minimum results for pH on a monthly basis and orthophosphate on a quarterly basis. The top and bottom of the lines represents the maximum and minimum values. The black dash in the middle of the line represents the average value.
https://www.dcwater.com/about-drinking-water-quality